Background
Audits in the pharmaceutical sector are a regulated and complex process. It involves a great deal of administrative effort and coordination. This is mainly due to the triangular relationship between the pharmaceutical company, the supplier and the audit service provider.
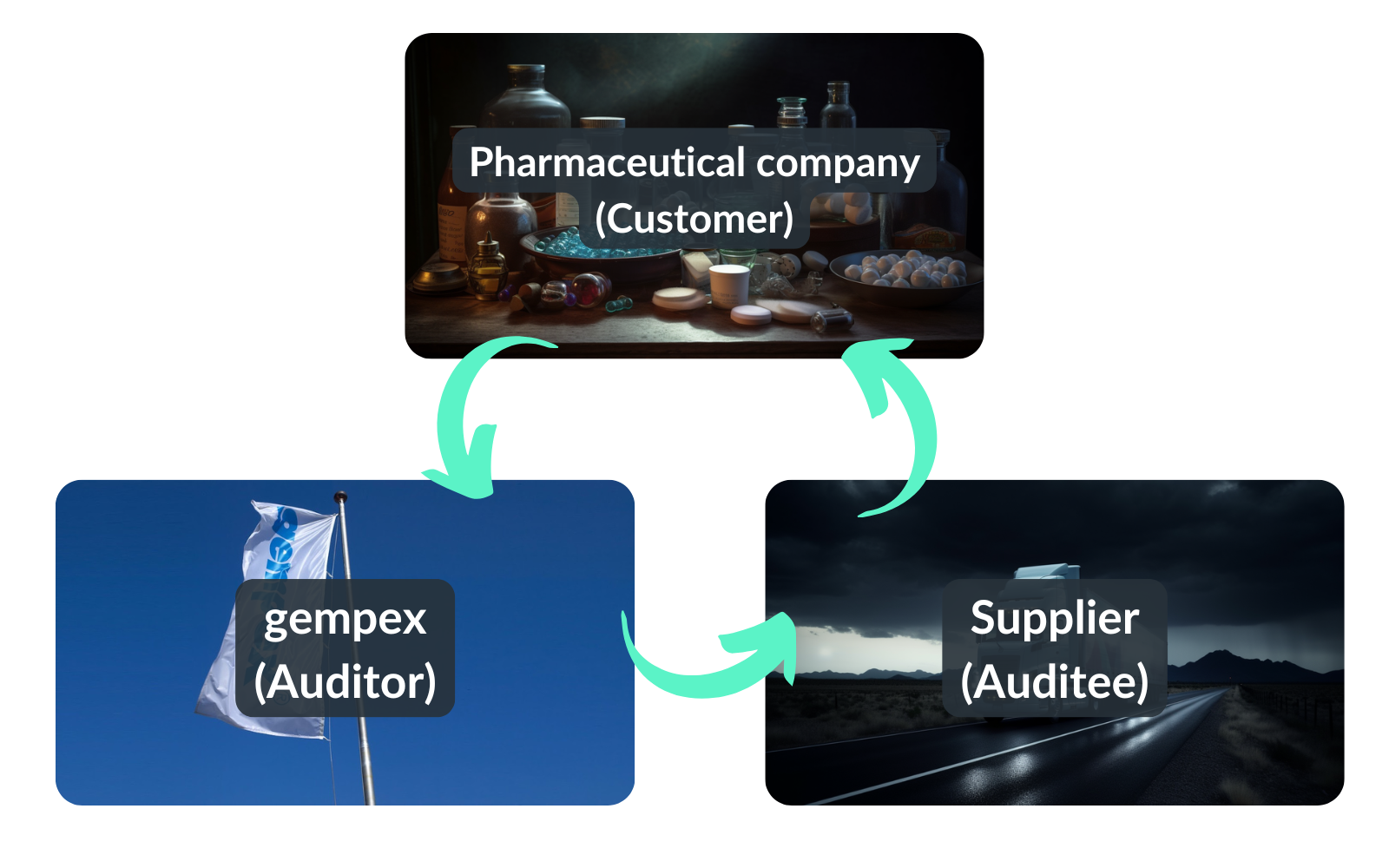
The pharmaceutical company (our customer gempex) ensures that the supplier's production meets all quality standards. The supplier (audit) supplies the pharmaceutical company and must meet those quality standards. gempex, as an audit service provider (auditor), is commissioned by the pharmaceutical company to carry out an audit. This ensures that the supplier adheres to the quality standards.
There is currently a lot of process knowledge in the minds of the employees at gempex. The documentation of various process steps and the storage of the relevant documents takes place in different locations:
- Email to communicate relevant information
- Excel files for documenting process steps
- SharePoint for document storage of individual steps
What gempex needed was a tool to reduce the coordination effort. It should also ensure more process transparency. In collaboration with SIDESTREAM, the audit tool was created - a central application to digitize the audit process end-to-end. On the one hand, the goal is to achieve efficient process design internally using the tool; on the other hand, it should also offer pharmaceutical companies and suppliers more transparency and better coordination.
The solution approach
Although the process always follows the same pattern with small variations, we have explicitly decided against developing a rigid tool that attempts to replicate the process 1:1. Why? If we try to pour complex processes 1:1 into a tool, we run the risk that not every small process adjustment or peculiarity will be reflected and can only be solved by adapting the tool. Instead, we pursued a flexible approach using generic task blocks and definable process steps. The task blocks are the following:
- Upload a document
- Choosing a date
- Confirmation of a completed task
- Entering text
Collaboration
Based on the initial input and requirements from gempex, we carried out a concept sprint. In close communication with gempex, we developed a profound understanding of the audit process. Based on this, we created a UI mock for the tool as well as a detailed list of planned features and work packages.
The implementation took place in two-week sprints. We developed new features and at the same time the gempex employees were able to test the latest version of the tool on a test environment and give feedback.
This made it possible to quickly create a first usable version. This covered the audit process end-to-end and already represented an improvement to the existing solution consisting of Excel and Sharepoint. The roll-out for different user groups took place gradually:
- initially for the coordinator (gempex-internal: person primarily responsible for all audit processes)
- then for the auditor (internal to Gempex: person carrying out the audit)
- then for the customer (gempex’s customer: pharmaceutical company) and the auditee (gempex’s customer’s supplier)
Technology Deep Dive
The audit tool is a container-based fullstack Typescript application. It is hosted in a Kubernetes cluster from AWS. This enables access from anywhere with high security standards and good scalability.
User management is done via GoTrue and a self-developed UI. This keeps user and rights management intuitive from within the tool. Dedicated IT support is not necessary.
PostgreSQL and Prisma ORM were used for the database. This allowed us to achieve a higher development speed and lower error-proneness through Prisma's Schema Definition Language (SDL).
The event tracking and sending of email notifications are based on Graphile-Worker and Prisma ORM middleware.
For a modern user interface we used VueJs and AntDesign.

.svg)



